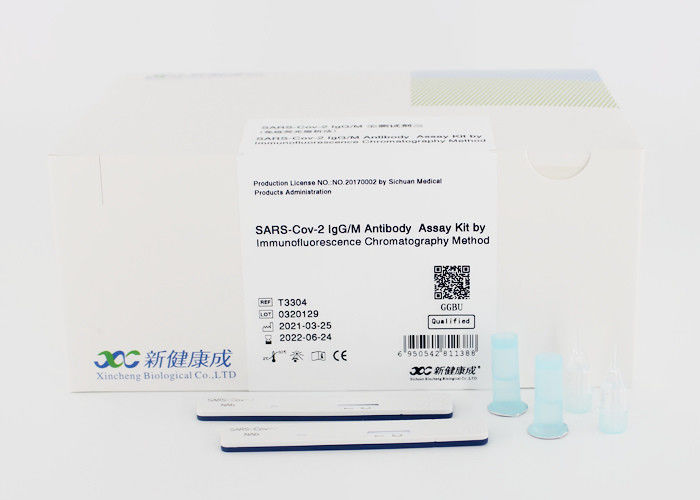
POCT With Blood for SARS-CoV-2 IgG, IgM Antibody Assay Kit
INTENDED USE:
SARS-CoV-2 IgM/IgG Antibody Test Kit is a single-use rapid immunochromatographic test for the qualitative detection and differentiation of Immunoglobulin M(IgM) and Immunoglobulin G(IgG) antibodies to SARS-CoV-2 in human serum, plasma (heparin, dipotassium EDTA, and sodium citrate), and venous whole blood (heparin, dipotassium EDTA, and sodium citrate). The Biohit SARS-CoV-2 IgM/IgG Antibody Test Kit is intended for use as an aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection. At this time, it is unknown for how long antibodies persist following infection and if the presence of antibodies confers protective immunity. The Biohit SARS-CoV-2 IgM/IgG Antibody Test Kit should not be used to diagnose acute SARS-CoV-2 infection. Testing is limited to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. §263a, that meet requirements to perform moderate or high complexity tests. Results are for the detection of SARS-CoV-2 antibodies. IgM and IgG antibodies to SARSCoV-2 are generally detectable in blood several days after initial infection, although the duration of time that antibodies are present post-infection is not well characterized. Individuals may have detectable virus present for several weeks following seroconversion. Laboratories within the United States and its territories are required to report all positive results to the appropriate public health authorities. The sensitivity of Biohit SARS-CoV-2 IgM/IgG Antibody Test Kit early after infection is unknown. Negative results do not preclude SARS-CoV-2 infection. If acute infection is suspected, direct testing for SARS-CoV-2 is necessary. False positive results for Biohit SARS-CoV-2 IgM/IgG Antibody Test Kit may occur due to cross-reactivity from pre-existing antibodies or other possible causes. Due to the risk of false positive results, confirmation of positive results should be considered using a second, different IgG or IgM assay. The Biohit SARS-CoV-2 IgM/IgG Antibody Test Kit is only for use under the Food and Drug Administration’s Emergency Use Authorization.
WARNINGS AND PRECAUTIONS:
1. For prescription use only. For in vitro diagnostic use only. Do not use after expiration date.
2. This test has not been FDA cleared or approved; this test has been authorized by FDA under an EUA for use by laboratories certified under CLIA, that meet requirements to perform moderate or high complexity tests.
3. This test has been authorized only for the presence of IgM and IgG antibodies against SARS-CoV-2, not for any other viruses or pathogens.
4. This test is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostic tests for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or revoked sooner.
5. Samples for human serum, plasma or whole blood should be considered as potentially infectious. Operators should wear protective clothing, masks, gloves and take other appropriate safety precautions to avoid or reduce the risk of infection.
6. This test should be performed at 18 to 30℃ (64 to 86°F). If stored refrigerated, ensure that the pouch and buffer are brought to operating temperature before performing testing.
7. Follow the instructions for use carefully. Reliability of assay results cannot be guaranteed if there is any deviation from the instructions in this package insert.
8. Professionals must handle the potentially contaminated materials safely according to local requirements.
9. Do not smoke, drink, eat, or use cosmetics in the working area. Wear Personal Protective Equipment (PPE) and disposable gloves when working with samples and reagents. Wash hands after operations.
10. Wipe and wash any splashed sample with highly effective disinfectant. Avoid splashing and the formation of aerosols.
11. Use a new clean disposable sample dispensing plastic dropper or tip for every sample to avoid cross contamination.
12. Decontaminate and dispose of all samples, reaction kits, and potentially contaminated materials as if they were infectious waste, in a biohazard waste container.
13. Once the Cassette is removed from the pouch, Use the Cassette as soon as possible to avoid being humidified. The Cassette is sensitive to humidity as well as to heat.
14. Do not use the Cassette if the pouch is damaged or the seal is broken.
Parameter:
| Item |
Reaction Time |
Valid period |
RSample Type |
Storage |
| Parameter |
8 Minutes |
15 Months |
Serum/Plasma/Whole blood |
2~30°C |



 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!